Protocol: Immunoprecipitation

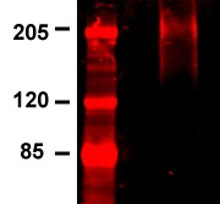
NOTE: This procedure may be used for cells labeled with radioactive compounds such as amino acids or orthophosphate. (Radioisotope use and disposal should conform to institutional and governmental regulations.) Cell labeling should be carried out in medium lacking the relevant nonradioactive compound. Starving cells appropriately prior to labeling is recommended. Incubate cultured cells (80–90% confluent monolayer in 100 mm cell culture plate or approximately 2–5 x 107 suspension cells in flask).
Example: Following starvation, remove culture medium and replace with methionine-free medium containing 5% dialyzed fetal calf serum and 100 µCi/ml 35S-methionine. Incubate 1 hour at 37º C. For some proteins a longer labeling period (up to 18 hours) is preferable. Wash cells with PBS (Buffers and General Solutions) as necessary to remove unincorporated 35S-methionine.
• Add 1–3 ml ice cold RIPA buffer (sc-24948) to subconfluent cell monolayer and incubate at 4º C for 10 minutes. For suspension cells, add the RIPA buffer to washed cell pellet in a microcentrifuge tube.
• Disrupt cells by repeated passage through a 21-gauge needle or sonication. Transfer to a microcentrifuge tube.
• Optional: Wash cell culture plate with additional 1.0 ml ice cold RIPA buffer and combine with original extract.
• Pellet cellular debris by centrifugation at 10,000xg for 10 minutes at 4º C. Transfer supernatant to a fresh microcentrifuge tube on ice.
• Preclear lysate by adding 1.0 µg of the appropriate control IgG (corresponding to the host species of the primary antibody), together with 20 µl of appropriate suspended (25% v/v) agarose conjugate (Protein A-Agarose, Protein G-Agarose, Protein A/G-Agarose, or Protein L-Agarose). Incubate at 4º C for 30 minutes.
• Centrifugation at 3,000 rpm (e.g. Eppendorf 5415D; approximately 1,000xg) for 30 seconds at 4º C.
• Transfer the supernatant, or approximately 100–1000 µg total cellular protein, to a microcentrifuge tube. Add 1–10 µl (i.e., 0.2–2 µg) primary antibody (optimal antibody concentration should be determined by titration) and incubate for 1–2 hours at 4º C. Alternatively, if antibody agarose conjugate is employed, add 20 µl (i.e., 5 µl packed beads) and incubate at 4º C for 1 hour to overnight with mixing; skip the next step.
• Add 20 µl of the appropriate agarose conjugate suspension (Protein A-Agarose, Protein G-Agarose, Protein A/G-Agarose, or Protein L-Agarose). Cap tubes and incubate at 4º C on a rocker platform or rotating device for 1 hour to overnight.
• Collect immunoprecipitates by centrifugation at 3,000 rpm (approximately 1,000xg) for 30 seconds at 4º C. Carefully aspirate and discard supernatant.
• Gently wash pellet 2–4 times with 1.0 ml RIPA buffer (more stringent) or PBS (less stringent), each time repeating centrifugation step above.
• After final wash, carefully aspirate and discard supernatant and resuspend pellet in 40 µl of 2x electrophoresis sample buffer (sc-24945).
• Boil samples for 2–3 minutes and subject to electrophoresis and autoradiography. Unused samples may be stored at -20º C.
Optional: After boiling, samples may be centrifuged to pellet the agarose beads followed by SDS-PAGE analysis of the supernatant.
